In this guide to Minnesota forage legumes, you’ll find features, uses and varieties for several common species, including various clovers, birdsfoot trefoil, crown vetch, cicer milkvetch and alfalfa.
Identifying perennial legumes
To identify individual legume species, you can use leaf traits.
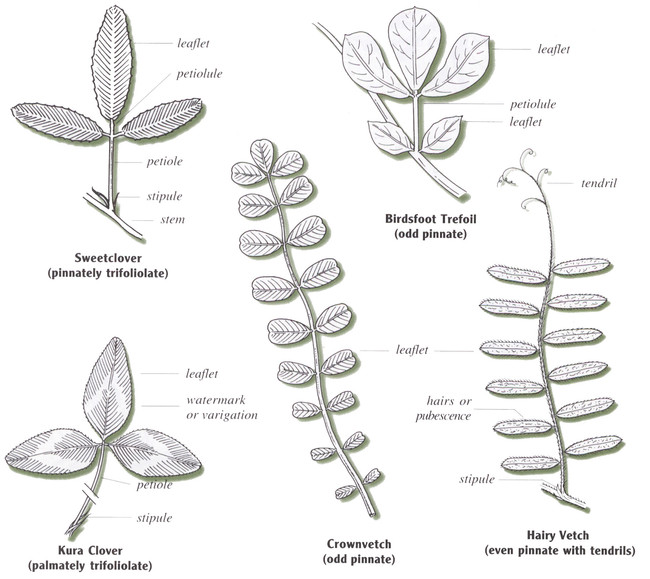
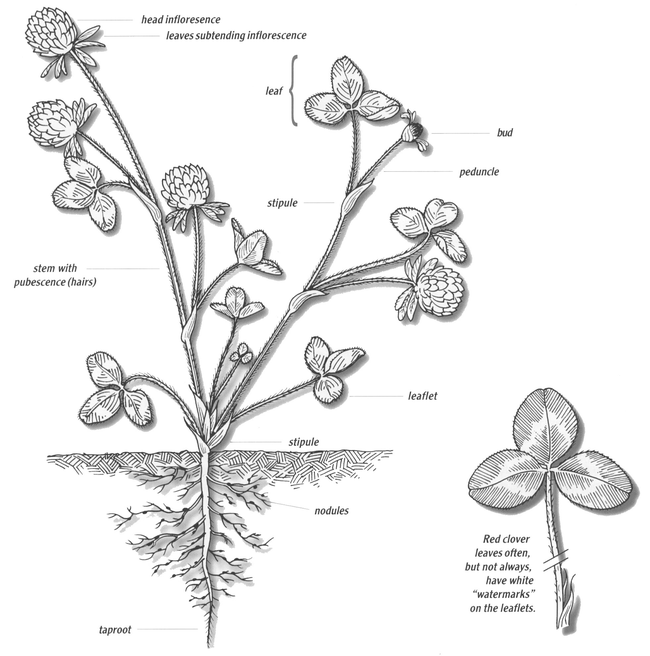
Legume leaves are usually compound, meaning they have more than one leaflet per leaf, and often have large stipules. The leaves are borne on petioles, which are attached to stems (Figure 1).
Leaflets per leaf
Though leaves of clovers and alfalfa typically have three leaflets per leaf, they sometimes have four or five.
The frequency of four or more leaflets per leaf is influenced by both the plant’s genetic makeup and the growing environment. Because four-leaf clovers occur relatively infrequently, they’re said in folklore to impart good luck. Some legumes have 10 or more leaflets.
Leaflet arrangements
Four arrangements, or organizations, of leaflets occur in the leaves of legume species commonly grown in the north-central region of the United States:
-
Palmately trifoliolate: Red, white, alsike and kura clover. These legumes are called true clovers.
-
Pinnately trifoliolate: Alfalfa and sweetclover.
-
Odd pinnate: Birdsfoot trefoil, crownvetch and cicer milkvetch.
-
Even pinnate with tendrils: Hairy vetch.
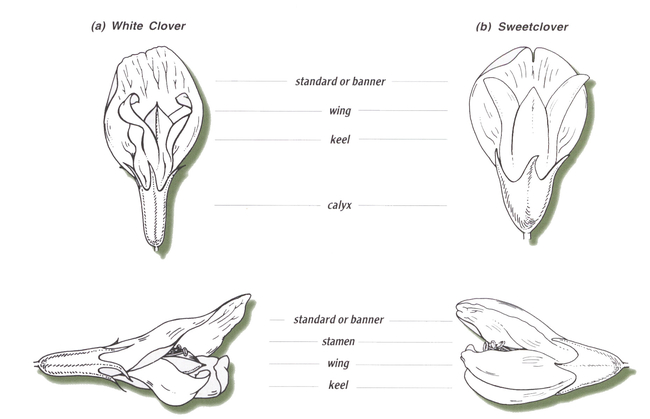
Legume flowers usually are showy and colorful. These features enhance the plants’ ability to attract its insect pollinators, including many species of native bees as well as the European honeybee.
Flower parts
Legume flower parts are the standard (also called the banner), wings and keel (Figure 2). The keel surrounds the male and female sexual parts.
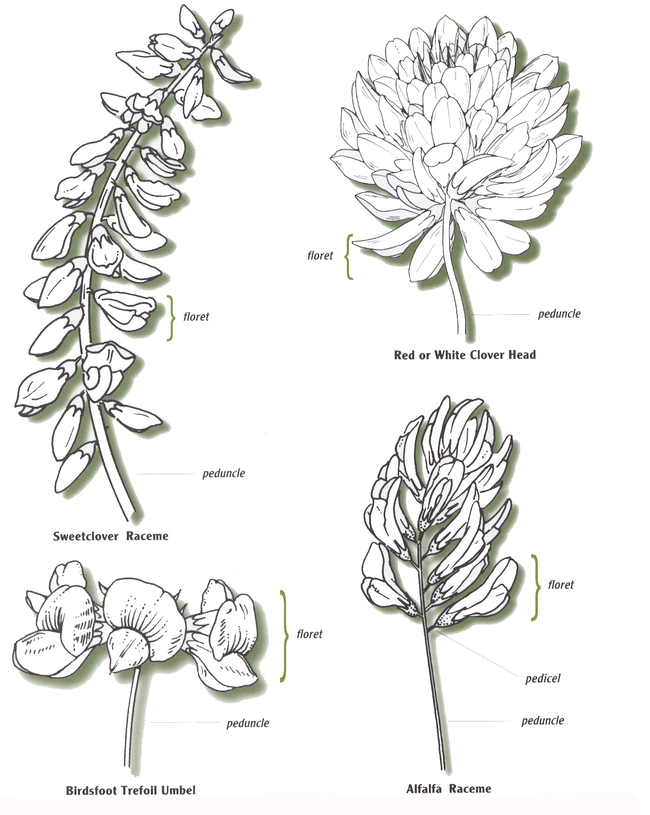
Inflorescences
Legume flowers are arranged in groups called inflorescences (Figure 3). The most common legume inflorescences are the:
-
Head: In red, white, alsike and kura clover.
-
Raceme: Alfalfa, sweetclover and cicer milkvetch.
-
Umbel: Birdsfoot trefoil and crownvetch.
A head typically will contain many flowers, while racemes and umbels contain few.
Pods
After pollination, legume seeds develop in pods. Pods can contain several seeds, as in birdsfoot trefoil and alfalfa, or only one seed, as in kura clover and sweetclover. When pods are dried, they can shatter and seed is dispersed.
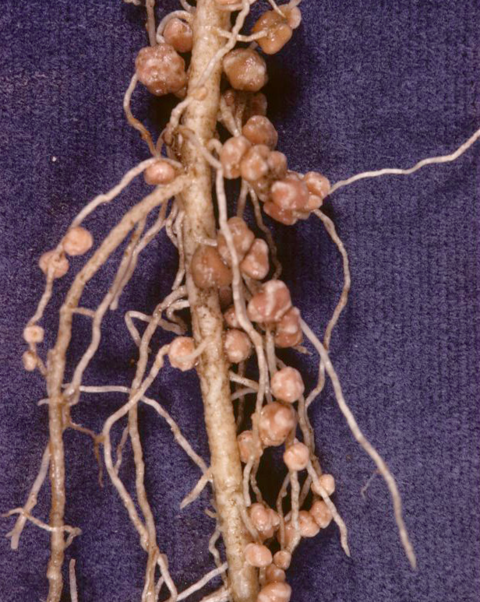
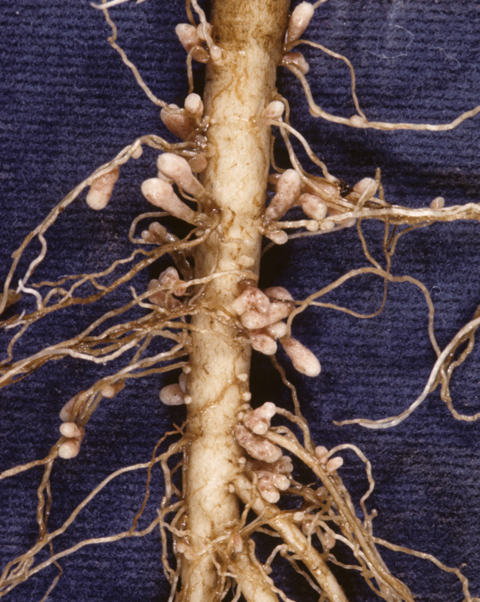
Forage legumes are usually tap-rooted plants with fine secondary roots produced from the tap root. These secondary roots are usually nodulated by nitrogen-fixing bacteria. Figures 4 and 5 illustrates this for birdsfoot trefoil and red clover.
A very large tap root gives legumes such as alfalfa, kura clover and sweetclover greater drought tolerance than other forage legumes. In contrast, the more fibrous, shallow root systems of other legumes, such as white and alsike clover, reduce their drought resistance.
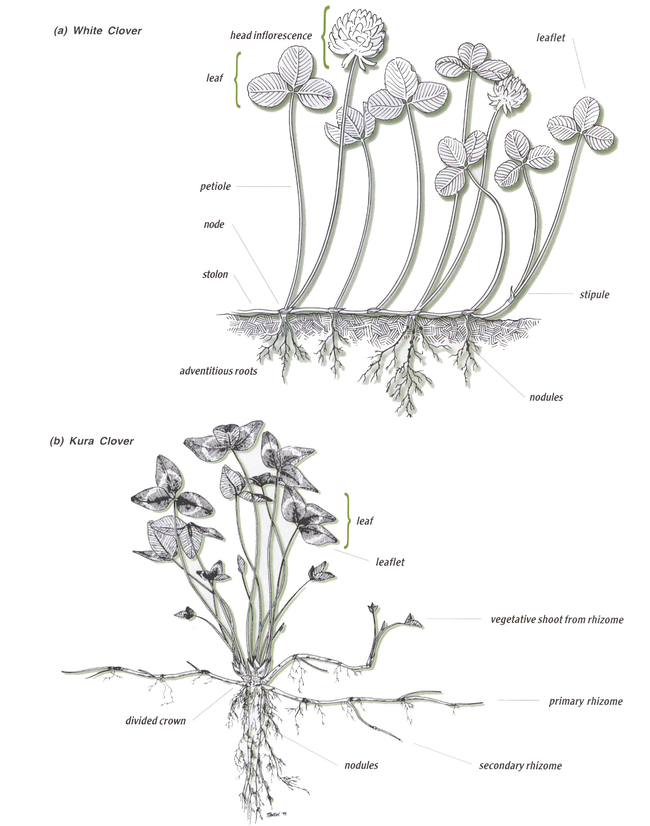
Stolons are horizontal aboveground stems (Figure 6). Rhizomes are horizontal belowground stems.
Both allow for vegetative reproduction without seeds, as new stems and roots can arise from nodes on stolons and rhizomes. This enhances plant persistence while creating more root sites for nodule growth.
Stolons are found in white clover, while rhizomes are found in kura clover, cicer milkvetch and crownvetch. Legumes with rhizomes are among the most persistent species.
Forage legumes
Kura clover (Trifolium ambiguum Bieb.) is a relatively low-growing, spreading perennial with excellent potential for grazing. It’s also called Caucasian, Pellett’s or honey clover.
History
Kura clover is native to the Caucasus region of Europe and named for the Kura River in the country of Georgia. It was introduced into the United States in about 1910, but remained little known until the 1940s.
That’s when Frank Pellett, impressed with its potential for honey production and its desirable agronomic traits, wrote about it in the American Bee Journal. However, interest didn’t increase until recently because of inadequate seed supplies of improved varieties and the unavailability of appropriate nitrogen-fixing rhizobium.
Varieties
The Soil Conservation Service and the University of Kentucky released the first-named U.S. variety, Rhizo, in 1990. Very persistent, it’s survived more than 15 years of continuous grazing in St. Paul and 12 years mixed with different grasses in Arlington, Wis.
Newer varieties include Everlast, Endura and Cossack. Endura has survived 22 years in mixture with tall fescue and smooth bromegrass in rotationally grazed pastures near Lancaster, Wis.
Characteristics
Kura clover has a deep branching taproot and rhizomes (horizontal belowground stems), which enable it to vigorously spread (Figure 6). Its crowns can be two inches below the soil surface. Individual plants can increase through rhizome growth by about one foot per year with no competition, and less with grass competition.
By fall of the seeding year, kura clover can have significant root and rhizome growth. A five-year-old stand can produce more than three tons per acre of belowground biomass (30 percent roots, 45 percent rhizomes, 25 percent crowns).
Leaflets are usually trifoliolate, oblong and watermarked, with considerable variation in leaf characteristics in a population. Leaves with four or five leaflets have been observed. Leaflets and stems are not hairy, but leaflet margins are acutely serrated at the edges.
Leaf size varies considerably with growing conditions during the season, spanning from 1 to 3 inches long and 1/4 to 2 inches wide.
Adaption
Kura clover has excellent tolerance to many stressful management and climatic factors. It has no major disease problems and is productive in diverse environments. Kura clover has greater persistence under rotational and continuous grazing, and frequent cutting, than any commonly grown legume.
Following two to four cuts per year for three years in southern Minnesota, kura clover had plant populations greater than 90 percent (alfalfa and other legumes were 50 percent or less). Its persistence is due in part to the extensive underground root, crown and rhizome system that’s a site for considerable carbohydrate storage.
For legumes like alfalfa and red clover, frequent harvesting depletes root carbohydrate concentration, but concentration of carbohydrates in kura clover’s belowground structures is only minimally affected.
Tolerance to weather
Kura clover is very winter-hardy. It goes dormant in the early fall in response to the short day length and low temperatures. It’s very resistant to injury from freezing and thawing, persisting more than 20 years and surviving extreme winter conditions in Minnesota and Wisconsin, while all other legumes died.
Kura clover has excellent tolerance to drought, although it’ll become dormant during extended dry periods and yield less than alfalfa (but similar to other clovers and birdsfoot trefoil). Although herbage growth reduces during drought, kura clover resumes growth following replenishment of soil moisture.
It can also often withstand poorly drained soils, survive flooding and survive on sites with a high water table. In Australia, kura clover plants had an 80 percent survival rate when flooded up to 40 days.
Uses
Kura clover is best-suited as a grazing crop because of its prostrate growth habit and very leafy, high-moisture forage. However, its first growth in the spring that contains an elongated stem can be harvested for hay or haylage. Subsequent regrowth will be leaves supported by petioles originating from crowns and rhizomes.
Consequently, for most of the season, kura clover is very leafy and high in feeding value. Its forage quality often exceeds the quality of other commonly grown legumes. Greatest yields occur in the spring; yields are less in summer and fall. Forage yields range from two to six tons per acre, with an average of about four tons per acre being most likely.
Grazing mixtures
Mixtures of grass and kura clover can produce quality forage suitable for lactating dairy cows or to support high weight gains of lambs or steers. Kura clover tolerates rotational grazing at intervals of 14 to 28 days or, though not recommended, continuous grazing.
Under stressful grazing or clipping, plants adapt by shortening leaf petioles, resulting in leaves closer to the soil surface. Unlike red clover, breeding ewes can safely graze kura clover because it doesn’t contain phytoestrogens.
Because of very low fiber content, high protein and potential for bloat, plant kura clover in mixture with perennial grasses. Mixtures should be 30 to 60 percent grass. Kura clover can successfully be established with most commonly used perennial grasses.
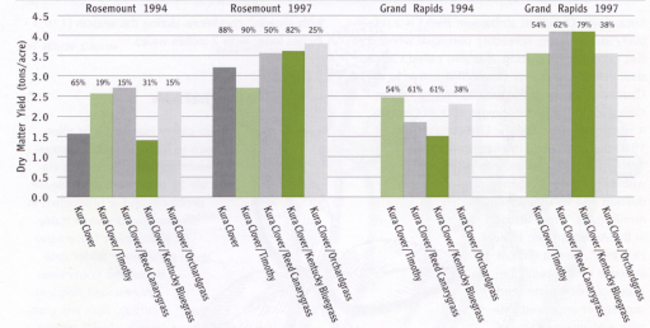
Base your choice of grass species on productivity and persistence of the grass at a site, and on management preference for any particular grass. Long-term grazing trials in Minnesota and Wisconsin show that grass selection influences the yield and grass composition of mixtures (Figure 8).
Mixtures of well-nodulated kura clover with grass have yielded as much as grass fertilized with up to 300 pounds of nitrogen per acre. Establishing kura clover with the noncompetitive birdsfoot trefoil enhances seedling-year yield, first-year yield and kura clover establishment. Over time, kura clover population increases while birdsfoot trefoil population declines.
Establishment
Establishing kura clover is more challenging than most other forage legumes. It has less seedling vigor than white clover and birdsfoot trefoil. Seedlings are fragile and develop slowly.
However, resources spent to establish kura clover are an investment that provides years of returns. Forage production and stand density in the seeding year will usually be low but, as it’s been said, “Kura clover sleeps in the first year, creeps in the second year and leaps in the third year.”
Weed control
Because of its lack of seedling vigor, it’s essential to minimize competition with weeds or companion crops at establishment. To achieve the greatest kura clover establishment yields, use an herbicide for weed control in the seeding year. If you plant companion crops to reduce soil erosion, harvest them for forage at vegetative stages to reduce competition.
Kura clover has also been successfully seeded into pastures with no-till strategies, provided that existing grasses are suppressed with herbicides such as glyphosate. No-till approaches reduce soil erosion and can reduce annual weed pressure.
Transplanting
Kura clover has been successfully established by transplanting portions of the underground rhizomes. Transplanted rhizomes with one or more nodes can form new roots and crowns and spread within a field if soil moisture and fertility are adequate.
Large fields have been established using a potato digger to extract rhizomes and crowns followed by spreading of the plant material onto a newly tilled field using a manure spreader and incorporating by disking.
Fertilizers
Biological nitrogen fixation develops more slowly for kura clover than for other legumes.
Improve establishment and seeding year yields by applying small amounts of nitrogen fertilizer in the seeding year on coarse soils low in nitrogen (20 pounds of nitrogen per acre at establishment and again at first harvest, approximately 60 days later). However, nitrogen fertilization can sometimes also increase weed competition if weeds aren’t controlled.
Soil stabilization
Because of its prostrate and spreading growth habit, kura clover has potential for use in soil stabilization projects. Its dense rhizome mat holds soil and prevents erosion.
With adequate suppression and seeding technology, kura clover can also be managed as a living mulch in corn with little or no reduction in grain or silage yield.
Kura clover supplies almost all the nitrogen required, provides permanent ground cover to reduce soil and nutrient runoff and recovers to full production of pasture the following season. However, when soybean was no-till seeded into a suppressed kura clover living mulch, soybean yields were reduced when compared to soybean seeded into kura clover killed by tillage.
Seed production
Kura clover flowers in response to long days in the spring.
Initial regrowth is upright stems supporting one or two large, fragrant, pink-white flowers. If the initial regrowth isn’t cut and allowed to mature, seed is produced in July or August.
Producing kura clover seed is more difficult than for many other legume forage species. Growers should consider a field’s previous crop history. Seed purity is an issue because legumes previously on the site, such as red clover, produce a similarly sized seed as kura. This makes them difficult to separate.
Planting and growing
Seed production fields are seeded in rows, or broadcast, at approximately one-quarter of the forage seeding rate. A firm seedbed allowing a uniform seeding depth of 1/4 to 1/2 inch is desirable. Kura clover is generally established using herbicides, without a companion crop, after the initial spring weed flush, and before late July.
Soil fertility requirements for seed production are the same as for forage production; a soil test prior to establishment and every two years during production is recommended. After establishment, rogueing or spot-spraying to eliminate problem perennial weeds such as thistles may be necessary.
Pollinating insects, especially bees, are important for seed production. One honeybee hive per acre is recommended for seed production, but you may need more bees if there’s competition for pollinators from other crops.
Harvesting
Successful harvesting requires prompt, timely and careful action. Swath fields when the majority of stems have turned brown, and combine when the crop is dry or direct-combine in the field after using a chemical desiccant.
To maximize seed yield, careful combine calibration is required because the seed is difficult to thresh. Kura clover seed fields may become sod-bound after several years due to the large root mass and rhizome production. Decline in seed production may follow, which would necessitate taking the field out of production.
An additional use associated with seed production is honey production. Kura clover flowers are highly scented with a shallow corolla, keeping its high sugar content nectar readily available to bees. If kura clover is allowed to flower, you can manage kura clover pastures or seed production fields to produce honey as an additional source of income.
History
Red clover (Trifolium pratense L.) is native to Asia and southern Europe, and is the most important and widely distributed of the clovers. Its name is likely derived from its blossom’s deep red color.
Red clover was introduced into England in 1650 and apparently into North America shortly afterwards by early colonists. It’s used in regions where soil pH is less-than-optimum for alfalfa and where forage yield goals are lower.
Varieties
There are two general types of red clover. Most varieties grown in the northern regions of the United States are medium, also called multiple cut types. The other type, mammoth red clover, is later-flowering than medium red clover and produces only one crop of hay per season.
Red clover has pubescent (i.e., hairy) upright growing stems that originate from a narrow crown near the soil surface (Figure 9). The plant has a taproot with many side branches, but its roots do not penetrate the soil to the depth of sweetclover, kura clover and alfalfa.
Adaption
Red clover is a short-lived perennial, which usually persists only two or three years. Breeders have improved red clover’s resistance to diseases and the persistence of many varieties. New varieties are available, but all can have their stand life shortened by winter injury.
Red clover’s persistence is reduced by high temperature, low moisture and flooding. Red clover is susceptible to winter injury partially because the plant’s crown is at the soil surface and not beneath it like alfalfa and kura clover crowns. In areas without adequate snow cover, red clover may only live one year.
Red clover is adapted to a wide range of soil types, except those in areas prone to drought. It tolerates a pH as low as 5.5. In northern sites such as Grand Rapids, red clover under two-cut harvest management will have similar yields as alfalfa. With more intensive cutting schedules, yield will be less.
Use
Red clover is used for hay, silage and pasture. It’s often used as a hay and pasture crop alternative to alfalfa on heavy soils of low pH in northern Minnesota. In southern Minnesota, it generally yields less and is less persistent than alfalfa, partly due to its low drought tolerance.
Red clover produces high-quality forage due to its leafiness and stems, which are relatively high in nutritive value. It also has somewhat greater undegradable protein and greater stem and fiber digestibility than alfalfa. Red clover has enzymes that result in less protein breakdown to non-protein nitrogen during ensiling.
Red clover is frequently planted in mixtures with grasses to minimize ruminant bloat during grazing and to enhance hay-drying. It contains phytoestrogens that reduce conception rates of ewes that have consumed red clover pasture or silage, but this apparently hasn’t been an issue with cattle.
Pasture
Animal performance on red clover pasture is similar to their performance on alfalfa, but product per acre is less. However, the long-term carrying capacity and production per acre is lower for red clover than for alfalfa due to less stand persistence.
Red clover has excellent seedling vigor and is good for pasture renovation using sod-seeding or frost-seeding.
Hay and silage
Red clover is a two- or three-cut crop that’s usually harvested at early flowering (when about 25 percent of the stems are flowering). Allowing the crop to reach full flower (with 100 percent of the stems having mature flowers) will result in low-quality forage.
Because red clover is high in moisture, it’s also sometimes difficult to dry it to moisture levels low enough for storage as hay. Low-moisture silage is often made to reduce risk of rain damage.
Red clover hay may cause slobbering by livestock if the hay becomes infected by black patch disease. This disease is caused by Rhizoctonia leguminicola, which produces the alkaloid slaframine that induces salivation.
Seed production
Red clover also offers potential for on-farm seed production.
For multiple-cut varieties, the first crop in early June is harvested or clipped and seed is produced on the second crop. Seed yields increase dramatically if adequate pollinators such as honey bees are present.
White clover (Trifolium repens L.) is distributed throughout the world. It thrives in areas with fertile soils, good soil moisture and grazing animals.
As its name implies, white clover has white blossoms borne on long peduncles. White clover is unique among legumes because it spreads by stolons, its horizontal aboveground stems (Figure 6). Its herbage is usually shorter than other legumes because it has no upright stems. Its leaves and flowers originate from stolons on the soil surface.
History
White clover’s exact origin is disputed, but it most likely evolved either in the eastern Mediterranean region or in western Asia.
White clover was grown in England in the early 1700s and introduced into North America by early colonists. As land was cleared for farms and deforestation occurred in the colonies, grazing livestock and other animals rapidly spread the white clover’s small seed.
White clover isn’t a major cultivated forage legume in the north-central United States because it generally has poor persistence and low productivity. However, because of its prolific seed production, it’s widely distributed and found in lawns, pastures and waste areas. It’s also frequently included as a component of pasture mixtures.
Varieties
Three types of white clover grow in the north-central United States:
-
Ladino.
-
White Dutch, also known as intermediate or medium.
-
Wild white clover.
White Dutch and wild white clovers are very prostrate. They’re often found in permanent pastures and lawns. They profusely flower and reseed themselves. Although they have low forage productivity, these clovers contribute nitrogen that’s used by grasses in pastures.
Ladino white clover is a large type of white clover that’s more productive than the Dutch or wild white clovers, and is suited for forage production. Most white clovers sold in the United States are unnamed, common types because little variety development has occurred.
Merit 3, Shasta and Sacramento are older varieties that are sometimes available. More recently, the varieties Alice and Kopu II were developed in New Zealand for use in grazing systems. Kopu II has demonstrated superior stolon density and survival over a three-year period in Wisconsin.
Adaptation
White clover is adapted to soils and regions, which have a constant supply of moisture. It has a very shallow root system and no drought tolerance.
White clover is most productive during summers with cooler temperatures and well-distributed rainfall. It tolerates acid soils (pH 5.5) but not saline or alkaline soils.
White clovers are less winter-hardy than red clover. While they can overwinter, plants will not usually survive without adequate snow cover.
White clover primarily persists in pastures by regeneration from seed produced the previous year, although stolons can sometimes overwinter. Numerous diseases also reduce persistence.
Use
White clovers are most often used for pastures and sold as components of pasture mixtures, which include grasses. Recently, ladino white clover has been included as a component in wildlife mixtures sold to feed and attract whitetail deer.
White Dutch and wild white clovers often naturally occur in heavily grazed pastures and regenerate each year by reseeding. White clovers are good for frost-seeding into pastures in winter or early spring.
White clovers have high forage nutritive value because the forage consists mostly of leaves, but the forage can cause bloat. Ladino clover is the only white clover that grows tall enough to be cut for hay. Harvesting at early flowering is recommended.
Birdsfoot trefoil (Lotus corniculatus L.) is frequently used as a ground cover and its bright yellow flowers are often seen along highways during the spring and summer.
Birdsfoot trefoil has very small seeds and poor seedling vigor. Care must be taken to assure its establishment.
History
Birdsfoot trefoil is a native of Europe and Asia that may have been brought to the American colonies in soil used for ship ballast. Plantings were established in the late 1880s at several agricultural experiment stations in the eastern United States.
Characteristics
Birdsfoot trefoil derives its name from the claw-like arrangement of its seed pods which, to some, resembles a bird’s foot. The pods shatter and release their seeds when ripe, making seed harvest difficult.
Birdsfoot trefoil has fine stems that tend to lodge. It makes considerable regrowth from axillary buds on its lodged stems and is less dependent on regrowth from crown buds than alfalfa. Its leaves consist of five leaflets, three grouped together at the end of the petiolule and two at the leaf base.
Varieties
Two main types of birdsfoot trefoil are used for forage production in Minnesota.
Low-growing types such as Empire and Dawn are fine-stemmed and very prostrate. Both are also later in flowering and more winter-hardy than upright (also called European) types. Viking is an upright type.
Low-growing types of birdsfoot trefoil are best-suited for pasture. Upright types are adapted to both hay and pasture usage. They make more rapid regrowth but are less persistent than the low-growing types. Norcen is an intermediate type, which is persistent and well-adapted to growing conditions common in Minnesota.
The Minnesota Agricultural Experiment Station released two more recent varieties of birdsfoot trefoil, Neultin and Roseau, but they’re no longer produced.
Adaption
Birdsfoot trefoil is very tolerant of waterlogged soils and can withstand several weeks of flooding. It’s also tolerant of acid soils (pH 5.0), and is moderately tolerant of high alkalinity and salinity. On acid and poorly drained soils, birdsfoot trefoil will have greater yields than red clover and alfalfa.
Birdsfoot trefoil is adapted to most areas of Minnesota, but only has moderate drought and heat tolerance. It’s less winter-hardy than alfalfa.
Although birdsfoot trefoil is a perennial, when it’s intensively managed, forage yield and stands will usually persist for only three or four years due to disease and winter injury. Its yield and persistence are best when cut or grazed two to three times per season.
Achieve stand persistence by allowing the crop to flower and set seed. Pods shatter when mature, and seeds are dispersed by wind, water or grazing animals.
Use
Primarily consider birdsfoot trefoil for pasture systems where animals can graze the forage.
Grazing
Birdsfoot trefoil contains tannins, which prevent legume bloat in grazing animals. In addition, because microbes in the rumen (i.e., bypass protein) can less readily break down the protein in birdsfoot trefoil, ruminant animals use its protein more effectively than the protein in either alfalfa or red clover.
Although birdsfoot trefoil has a lower yield and carrying capacity than alfalfa, it can provide high daily gains for grazing animals.
Delayed grazing
Birdsfoot trefoil is a good legume to accumulate in place (i.e., stockpiling), delaying grazing until midsummer or late fall following a killing frost, when forage is usually in short supply. Because of its prostrate growth habit, birdsfoot trefoil can withstand heavy grazing.
Delayed grazing is possible with birdsfoot trefoil because it holds its leaves at maturity, retaining its forage quality. Delayed grazing also allows abundant seed production and natural reseeding.
This makes birdsfoot trefoil pasture a good component of a grazing system that uses cool-season grasses or other forages for early-spring grazing. This leaves birdsfoot trefoil grazing for the middle part of summer.
Hay
Birdsfoot trefoil is difficult to cut for hay because of lodging, but it can produce a high-quality hay crop. Hay harvests should occur at early flowering. Mechanical harvesting at 1- to 3-inch cutting heights for hay or silage will generally reduce stand life compared to grazing.
Seed birdsfoot trefoil with a grass like timothy to reduce weed invasion.
Other forage crop variety trials
Seed production
Birdsfoot trefoil is a prolific seed producer. In northern Minnesota, a seed industry has been established.
Alsike clover (Trifolium hybridum L.) volunteers in many permanent pastures and roadsides in areas of the region with adequate rainfall and good winter snow cover. It can be perennial under such ideal conditions, but in the north-central region, it typically behaves as an annual or biennial.
Alsike clover was once frequently used on poorly drained and acidic soils, when all forages were only infrequently harvested. However, it’s not currently a prominent forage legume.
Alsike clover is still used as a component of hay and pasture mixtures, with red clover and grasses such as timothy and red top, because of its low seed cost and adaptation to specific soil conditions.
History
Alsike clover is named after a location in Sweden where it was cultivated as early as 1750. It’s a distinct species, not a hybrid of red and white clover as was once thought.
Alsike clover is native to the temperate regions of Europe and Asia. Seed was distributed in the United States by the Patent Office in 1854, but it was probably introduced into the United States in about 1839.
Varieties
There are no released varieties specifically developed in the United States; most seed is common. Tetra is a tetraploid variety developed in Sweden. In Minnesota, it has similar yield and persistence as the common varieties.
Characteristics
Alsike clover has an upright growth habit like red clover. However, because its stems are fine, they usually lodge if alsike isn’t grown in mixture with a more upright grass or legume.
Alsike clover stems can grow 3 to 5 feet long if uncut, though stems are normally 1 to 2 feet long. Its leaves and stems lack pubescence. Leaflets are finely serrated along the entire edge and are never variegated or watermarked.
Alsike clover has white to pinkish flowers that originate in leaf axils up and down the stem. Flowers are similar in size to those of white clover. Alsike clover has an indeterminate growth habit that gives uncut stems the potential to simultaneously bear buds, flowers, immature seeds and ripe seeds along its entire length.
Seeds originating on flowers from lower portions of the stem may shatter before blooming on the stem’s upper portions ceases. This is why a large supply of hard seed is usually produced, contributing to natural reseeding in later years.
Adaptation
Alsike clover is best adapted to moist or poorly drained soils. It usually doesn’t persist on dry, sandy soils and isn’t tolerant of drought or high temperatures. It will tolerate waterlogged soils and can withstand spring flooding for up to six weeks.
Alsike clover will tolerate acidic soils with a pH of 5.0. Alsike clover is best adapted to the cool, moist climate of the northern parts of the United States’ north-central region, and to low-lying, moist areas in the region’s southern part. It can persist and be relatively productive on wet soils, acid and alkaline, where red and white clovers do poorly.
Use
Alsike clover is used for hay and pasture. However, its forage yield and persistence is low relative to other legumes, especially when grown on upland fertile soils and cut frequently. It’s best adapted to mixtures with grasses like timothy that are infrequently harvested, and harvested at full bloom.
For hay production, usually only one or two cuts are possible. The first spring growth that occurs under cooler conditions usually represents about 80 percent of the total season yield. Likewise, under grazing, expect the greatest yields in the spring, with little yield during summer grazing.
Forage often has higher quality and is leafier than forage of red or white clover. Alsike clover can cause bloat and photosensitization, making it best to grow in a mixture with grass.
History
Sweetclover is native to the Bakhara region of Asiatic Russia, and has been used as a green manure and a honey plant for more than 2,000 years. It was first reported growing in North America in Virginia in 1739.
It became recognized for its soil reclamation properties around 1900, when it successfully grew on many depleted, eroded soils of the southern United States.
Acreage planted to sweetclover has declined since usage peaked between 1925 and 1950. Several factors brought about its decline, including:
-
A decrease in the use of rotations.
-
The prevalence of cheap synthetic fertilizers.
-
A potential danger to ruminants from bleeding disease.
-
Damage by the sweetclover weevil (Sitona cylindricollis Fahr).
The sweetclover weevil is a dark green snout beetle about 3/16ths of an inch long. Adult weevils consume new seedlings and eat crescent-shaped areas from young leaves. Larvae injure the plant by attacking roots.
Today, a limited amount of sweetclover is grown on Conservation Reserve Program (CRP) acreage. Sweetclover is also commonly found on wasteland and on roadsides, where it regenerates by self-seeding. As the handbook “Forage and Pasture Crops” says, “sweetclover will grow anywhere, provided there is more than 17 inches of well distributed rain, and the soil is not sour.”
Characteristics
The name sweetclover is derived from the sweet odor of its crushed leaves. All sweetclovers contain coumarin, a bitter, stinging-tasting substance with a vanilla-like odor.
Coumarin is indirectly responsible for bleeding disease in livestock. In spoiled and molded sweetclover hay, coumarin transforms to dicoumarol, an anticoagulant. When cattle and sheep consume this spoiled hay, they develop bleeding disease. Horses rarely develop bleeding disease, but can develop colic from moldy hay.
While a cause of death in livestock, dicoumarol and its derivatives have saved many human lives by reducing blood clotting after surgery and the incidence of coronary thrombosis. Dicoumarol derivatives are also used in products like Warfarin for rodent control.
Sweetclover plants are tall, growing to heights of 2 to 4 feet, and have a thick, coarse stem. They produce large quantities of seed that shatters on maturity, leading to natural reseeding. Sweetclover leaves are pinnately compound with serrations around the entire leaf edge.
Most sweetclover varieties, being biennial, flower and die after their second year. There are white and yellow flowered types of sweetclover.
Varieties
Yellow sweetclover (Melilotus officinalis L.) flowers about two weeks earlier than white. The yellow types are smaller and lower-yielding, but also leafier and more drought-tolerant than white sweetclover (Melilotus alba L.).
Over time, a number of sweetclover varieties have been released. However, because of limited demand and on-farm seed production, variety integrity is often lost and seed of specific varieties may not be available.
Most sweetclover that’s found on the landscape usually contains about 2 percent coumarin, but some varieties with lower levels of coumarin have been marketed. Denta is a low-coumarin variety developed in Wisconsin.
In terms of biomass production, yellow and white varieties are similar. Yellow sweetclover makes less top growth in the fall of the first year than white varieties, but yellow increases its biomass yield with greater root growth. White sweetclover (also known as Bakhara, or Bakhara melliot) is taller and has a coarser stem than yellow sweetclover.
Most seed sold is common, however Evergreen (white blossomed) and Madrid (yellow blossomed) are two old-named varieties sometimes available. Hubam is an annual white-blossomed sweetclover variety that’s used as a green manure and emergency hay crop. It yields more forage, but produces less root biomass than the biennial sweetclovers.
Adaptation
Sweetclover requires nonacid (pH greater than 6.5), reasonably well-drained soils. It’s the legume best adapted to highly alkaline soils. It’s intolerant of poorly drained and flooded soils, but is drought-resistant and winter-hardy.
Use
Soil improvement
Sweetclover is one of the best legumes for soil improvement. It produces high yields of both herbage and root nitrogen, as well as organic matter when not cut. Before the advent of synthetic fertilizers, the Corn Belt routinely used sweetclover as a green manure crop.
When grown for green manure, biennial sweetclover is plowed under in the spring of the second year. Plowing in the spring kills the plants, provided plant growth is at least three inches. In contrast, with fall plowing, plants can regrow in the spring and become weedy.
Honey
Sweetclover is also an excellent source of high-quality honey. It produces an abundance of nectar, and the honey derived from it is light-colored and mild-flavored. One acre of sweetclover is sufficient for one hive of bees.
Harvested forage
Sweetclover isn’t a preferred legume for harvested forage. It’s tall-growing and stemmy, and the forage tends to be low in quality. When cut for hay, you can achieve yields of two to four tons per acre.
The best stage to cut sweetclover for hay is at bud to early blossom. A stubble of 8 to 12 inches is usually left to encourage regrowth. This is because regrowth comes from axillary buds on the stem instead of the crown.
Any cutting of biennial sweetclover the first year reduces root size and vigor the second year. Because of its high moisture content and its rank growth, curing is difficult.
Grazing
Sweetclover is low in palatability because of its coumarin content, and animals tend to eat other vegetation before eating sweetclover. Animals can, however, adjust to it. Sweetclover also causes bloat and scouring, and may taint milk.
Despite these limitations, grazing animals can perform well on sweetclover. Grazing may begin when plants are 14 inches tall, but maintain a minimum height of 8 inches to allow rapid regrowth. Plants become woody and unproductive if allowed to reach bud stage before initiating grazing.
Crownvetch has been extensively used for roadside stabilization and land reclamation, and for ornamental purposes. It hasn’t developed into an important forage legume in the north central region of the United States.
History
Crownvetch (Coronilla varia L.) is a native of middle and southern Europe. It became commercially available in the United States as an ornamental as early as 1890.
Interest in using crownvetch as a forage was generated by its discovery in a Pennsylvania field in 1935. It had been started from plants originally sown about 30 years earlier. The variety Penngift was derived from that population.
Characteristics
Crownvetch derives its name from its vetch-like leaves and the crown-like arrangement of its white-purple flowers in its inflorescence. Seeds are borne in nonshattering pods that break into sections when dry.
Crownvetch is indeterminate. Flowering and seed production continue through the entire growing season. It has a deep, creeping root system with a prostrate herbage growth habit.
Varieties
Penngift, Emerald and Chemung are the most widely used varieties.
Adaption
Crownvetch is tolerant of low fertility and low pH soils, but is best adapted to soils with a pH of 6 or above. It’s also drought- and disease-tolerant.
Crownvetch survives on roadsides in Minnesota where it’s not mowed and is protected from low winter temperatures.
However, don’t consider crownvetch for forage use in Minnesota. It lacks winter hardiness and has poor persistence when harvested. Crownvetch also has very poor seedling vigor and may require two years for successful establishment. Stands have been established by planting excised crowns, which are available in many nurseries.
Use
Crownvetch is best adapted for non-agricultural uses. However, if crownvetch is used as a forage, it’s best adapted for pasture because of its prostrate growth habit and non-bloating characteristic.
If harvested for hay, two harvests per season produce the greatest yields. Crownvetch hay contains nitro-compounds that can be toxic to nonruminants. These compounds also reduce its palatability to all grazing animals.
Cicer milkvetch (Astragalus cicer L.) has been evaluated and used for forage in the Great Plains and western United States. It has, however, only been evaluated in a limited number of Minnesota trials.
Cicer milkvetch is a vigorous, persistent, high-yielding legume that spreads by rhizomes and has a deep root system. Stands of cicer milkvetch have persisted 10 years under stressful conditions in Minnesota.
Characteristics
The herbage of cicer milkvetch is upright during regrowth, but because its stems can reach a length of 4 feet, they will lodge with maturity. Its leaves are pinnately compound with eight to 17 pairs of leaflets and a single terminal leaflet.
Yellow to white flowers are borne on racemes that originate in leaf axils. Its seed pods are bladder-like and can contain as many as 12 seeds.
Varieties
Cicer milkvetch has low seedling vigor and may take two years for good establishment. Lutana and Monarch are two varieties released in the United States.
University of Minnesota researchers developed a new variety, HiPal, that has improved palatability compared to other varieties. However, that seed is no longer available.
Adaption
Cicer milkvetch tolerates soil with slight acidity to moderate alkalinity. It’s intolerant of wet soils and flooding. Drought reduces forage yield, but stand persistence isn’t affected. Cicer milkvetch is very winter-hardy and disease- and insect-resistant.
Use
Cicer milkvetch has potential as a harvested or grazed forage crop.
Flowering is the recommended development stage when harvesting for hay. However, because of slow regrowth, it’s only a two- or three-cut crop when cut at the flowering stage. Forage nutritive value is high, often exceeding that of alfalfa.
Cicer milkvetch has a high carrying capacity when grazed, but sometimes provides lower average daily gains for livestock than most other legumes. This is due to unknown antiquality constituents, which reduce palatability (it’s not eaten by livestock when a choice is given) and cause photosensitization and hair loss for the grazing ruminants.
Alfalfa (Medicago sativa L.) is the most important and most widely planted perennial forage legume grown in the north-central United States and across the country. It’s the legume all others are measured against. We’re including alfalfa on this page primarily for comparison.
Alfalfa production for forage predominantly occurs in the north-central states of Minnesota, Wisconsin, North Dakota and South Dakota. However, seed is primarily produced in California, Idaho, Washington, Nevada and Oregon, where the climate and pollinators are optimum for good seed yields.
History
Alfalfa originated in southwestern Asia near modern-day Iraq, Iran and Afghanistan. It was described as an animal feed source as early as 490 before the common era, by the Roman writers Pliny and Strabo. It’s been dispersed throughout the world by explorers, armies and colonists.
Alfalfa is believed to have been originally introduced into the eastern North American colonies in 1736, with several additional early germplasm introductions occurring between 1840 and 1900.
Especially notable for northern states, such as Minnesota, was the introduction of a north-central region, winter-hardy variety in 1858 by Wendelin Grimm, an immigrant from Germany. Named for him, the variety Grimm was selected from his initial seeding in Carver County.
Varieties
Public institutions, such as land-grant universities, and private companies have had considerable success breeding alfalfa varieties.
This has produced many persistent, pest-resistant and high-yielding alfalfa varieties. In recent years, more than 100 alfalfa varieties have been marketed in Minnesota.
Alfalfa field crop variety trials
Characteristics
Most commercial alfalfa varieties have a deep tap root and upright herbage, which originates from a large crown. Its leaflets are serrated along half to a third of the leaf margin. Flowers are typically purple, but on some varieties, they may be yellow, white, cream or variegated across shades of blue and green.
Rooting depths of 20 to 30 feet are frequently reported for alfalfa. Herbage can reach a height of 3 to 4 feet if not cut. However, the crop will usually lodge before reaching that height. A few alfalfa varieties have creeping roots and rhizomes, which result in a more prostrate growth habit.
Adaptation
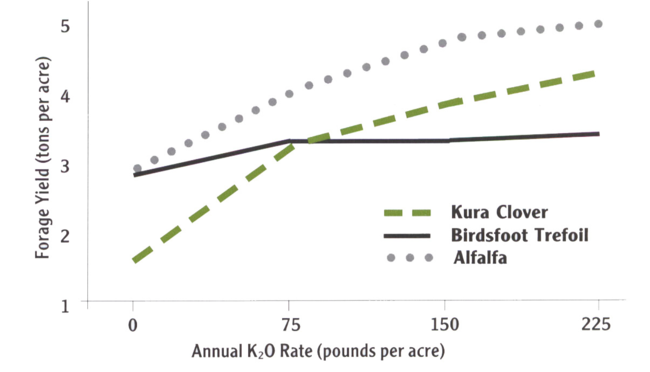
For good persistence and yield, alfalfa requires a soil pH of 6.5 to 7.0 and high levels of soil nutrients. Potassium is especially critical for high yields and persistence. Alfalfa is intolerant of flooding and water-logged soils.
Winter injury frequently affects the persistence of alfalfa and other perennials. Plant and environmental factors determine the extent of winter injury.
Plant factors affecting winter survival
During the winter, the alfalfa plant is somewhat like a hibernating animal. The plant has a low level of metabolic activity, and relies on stored carbohydrate reserves for energy during the winter and for regrowth in the spring.
Perennial portions of the plant – its crowns and roots – are dormant in the winter. In the spring, the plant regrows from crown buds formed the previous fall. Cold tolerance is greatest for crown buds and least for the roots.
Environmental factors affecting winter survival
Decreasing fall temperatures and shortening day lengths cause biochemical and morphological changes that allow the plant to tolerate low winter temperatures. While temperatures below 25 degrees Fahrenheit can kill alfalfa during the summer, very dormant and winter-hardy varieties can withstand temperatures as low as -5 in the fall.
However, most commonly grown hay varieties will experience winter injury at soil temperatures below 5. Prolonged, seven- to 10-day exposure to temperatures above about 45 can cause alfalfa to break dormancy and become susceptible to freezing injury. Dormancy is most easily broken by these temperature levels in late winter and early spring.
The soil, plant residue and snow protect alfalfa and other perennial plants from freezing winter temperatures. The plant’s first line of defense is to have a crown deeply buried by the soil. Varieties vary in this trait. Winter-hardy, grazing-tolerant types often have the deepest crowns.
Reducing the risk of winter injury
For the lowest risk of winter-injury, select varieties with a winter survivability rating of two and a fall dormancy of four. The National Alfalfa & Forage Alliance provides information on winter survivability – rated on a scale of one (most) to six (least) – for all marketed alfalfa varieties.
Plant residue, such as dried stems and leaves, protects the soil from rapid freezing and from freezing and thawing. It also catches snow that insulates the soil from low temperatures. To insulate the soil, you need a minimum snow depth of about 5 inches.
Alfalfa and all perennial legumes are intolerant of coverage by ice sheeting. Ice sheets can damage alfalfa crowns and cause carbon dioxide to build up around the plants.
Use
Alfalfa is used for hay, silage and pasture. In the north-central United States, small quantities are also dehydrated. Fields are often used for both grazing and hay production.
Because alfalfa’s forage quality potential is high, it’s often a key component of ruminant rations. Under good growing conditions, alfalfa produces greater yields of forage dry matter and protein per acre than any other commonly grown forage legumes.
Regional and local growing conditions, as well as the goals of the producer, all influence optimal harvest management decisions. Producer goals are usually based on the relative value of forage yield, forage quality and stand persistence in specific systems.
Forage quality
For example, a dairy producer in southern Minnesota who routinely four-cuts alfalfa at bud stage sacrifices dry matter or nutrient yield and stand persistence for excellent quality forage.
Such a forage has value as feed and replacement for high-cost energy and protein supplements. Furthermore, the increased forage quality from such a four-cut schedule can result in significantly more milk per acre than a three-cut schedule, according to the Alfalfa Management Guide.
Hay
If growing hay for market, where maximum tonnage is more valued than its forage quality, harvesting alfalfa at first flower may be the most profitable production strategy. You can harvest alfalfa grown for both feeding and marketing at different growth stages, depending on the specific circumstances of the livestock enterprise and market demand.
Grazing
When alfalfa is grazed, it results in good gains and carrying capacity. However, it does cause bloat in ruminants. Mixtures of alfalfa with grasses reduce the risk of bloat for grazing ruminants.
Crop rotation
Alfalfa is frequently grown in crop rotation with non-legumes such as corn. It can supply important nitrogen and organic matter for use by the subsequent crop.
The variety Nitro was specifically developed to supply both forage and nitrogen in crop rotations. It’s a non-dormant type that won’t consistently overwinter in Minnesota.
For more information on alfalfa management:
-
Alfalfa field crop variety trials - University of Minnesota
Affeldt, R.P., Albrecht, K.A., Boerboom, C.M., & Bures, E.J. (2000). Agronomy abstract from the International Meeting the American Society of Agronomy, the Crop Science Society of America, and the Canadian Society of Agronomy: Integrating herbicide resistant corn living mulch system (pp. 136). Madison, WI.
Baker, J.M. (2012). Vegetative propagation of kura clover: A field-scale test. Canadian Journal of Plant Science, 92, 1245-1251. https://doi.org/10.4141/cjps2012-014
Cumo, G.J., Johnson, D.G., & Head, W.A. (2001). Interseeding kura clover and interseeding kura clover and birdsfoot trefoil into existing cool-season grass pastures. Agronomy Journal, 93, 458-462. http://dx.doi.org/10.2134/agronj2001.932458x
DeHaan, L.R., Ehlke, N.J. & Sheaffer, C.C. (2001). Recurrent selection for seedling vigor in kura clover. Crop Science, 41, 1034-1041. http://dx.doi.org/doi:10.2135/cropsci2001.4141034x
Genrich, K.C., Sheaffer, C.C., & Elke, N.J. (1998). Kura clover growth and development during the seeding year. Crop Science, 38, 735-741. http://dx.doi.org/doi:10.2135/cropsci1998.0011183X003800030020x
Groya, F.L. & Sheaffer, C.C. (1985). Nitrogen from forage legumes: Harvest and tillage effects. Agronomy Journal, 77, 105-109. http://dx.doi.org/10.2134/agronj1985.00021962007700010025x
Hanson, A.A., Barnes, D.K., & Hill, R.R. Jr. (eds.). (1988). Alfalfa and alfalfa improvement. Agronomy Monograph 29. Madison, WI: American Society of Agronomy.
Laberge, G., Seguin, P., Peterson, P.R., Sheaffer, C.C. & Ehlke, N.J. (2005). Forage yield and species composition in years following kura clover sod-seeding into grass swards. Agronomy Journal, 97, 1352-1360. http://dx.doi.org/10.2134/agronj2005.0071
Meyer, D. (2005) Sweetclover production and management. (R-862; revised). North Dakota State University.
Mourino, F., Albrecht, K.A., Schaefer, D.M., & Berzaghi, P. (2003). Steer performance on kura clover-grass and red clover-grass mixed pastures. Agronomy Journal, 95, 652-659.
Musser, H.B., Hottenstein, W.L., & Stanford, J.P. (1954). Penngift crownvetch for slope control on Pennsylvania highways (Bulletin 576). Pennsylvania State University.
Pedersern, P., Bures, E.J., & Albrecht, K.A. (2009). Soybean production in a kura clover living mulch system. Agronomy Journal, 101, 653-656. http://dx.doi.org/10.2134/agronj2008.0073x
Peterson, P.R., Sheaffer, C.C., & Hall, M.H. (1992). Drought effects on perennial forage legume yield and quality. Agronomy Journal, 84, 774-779. http://dx.doi.org/10.2134/agronj1992.00021962008400050003x
Peterson, P.R., Sheaffer, C.C., Jordan, R.M., & Christians, C.J. (1994). Responses of kura clover to sheep grazing and clipping: I. Yield and forage quality. Agronomy Journal, 86, 655-660. http://dx.doi.org/10.2134/agronj1994.00021962008600040013x
Piper, C.V. (1924). Forage plants and their culture. New York: The MacMillan Company.
Seguin, P., Sheaffer, C.C., Ehlke, N.J. & Becker, R.L. (1999). Kura clover establishment methods. Journal of Production Agriculture, 12, 483-487. http://dx.doi.org/10.2134/jpa1999.0483
Sheaffer, C.C., Marten, G.C., Jordan, R.M., & Ristau, E.A. (1992). Forage potential of kura clover and birdsfoot trefoil when grazed by sheep. Agronomy Journal, 84, 176-180. http://dx.doi.org/10.2134/agronj1992.00021962008400020010x
Sheaffer, C.C. et al. (1989). Annual alfalfa in crop rotations. Minnesota Agricultural Experiment Station Bulletin (588-1989). St. Paul, MN: University of Minnesota.
Sheaffer, D.C., Mathison, R.D., & Seguin, P. (2008). Vegetative establishment of Kura clover. Canadian Journal of Plant Science, 88, 921-924. https://doi.org/10.4141/CJPS08026
Smith, W.K. & Gorz, H.J. (1965). Sweetclover improvement. Advances in Agronomy (17, pp. 163-231).
Townsend, C.E. (1985). Miscellaneous perennial clovers. In N.L. Taylor (ed.). Clover Science and Technology, Agronomy Monograph 25 (pp. 563-578). Madison, WI: American Society of Agronomy. http://doi.org/10.2134/agronmonogr25.c26
Undersander, D.J., Renz, M., Sheaffer, C., Shewmaker, G., & Sulc., M. (2015). Alfalfa Management Guide. American Society of Agronomy, Madison, WI.
Wheeler, W.A. (1950). Forage and Pasture Crops. New York: D. Van Nostrand Company, Inc.
Yost, M.A., Coulter, J.A., & Russelle, M.P. (2018). Managing the rotation from alfalfa to corn.
Zemenchik, R.A., Albrecht, K.A., Boerboom, C.M., & Lauer, J.G. (2000). Corn production with kura clover as a living mulch. Agronomy Journal, 92, 698-705. http://doi.org/10.2134/agronj2000.924698x
Reviewed in 2023