Since the 1920s, brown root rot (BRR) has been regarded as an important disease of forage legumes, including alfalfa, in the northern regions of North America.
Aboveground symptoms of brown root rot (BRR) consist of plants that are either slow to green up in the spring or die during the winter. In such instances of winterkill, stand losses often are assumed to exclusively be caused by exposure to harsh winter weather conditions.
However, we now know BRR contributes to plant death in Minnesota alfalfa stands in areas where Phoma sclerotioides (P. sclerotioides) – the pathogen that causes the disease – is present.
Disease symptoms
Substantial stand loss can occur if conditions support the disease's development. Losses either happen in a single winter season, or gradually over many years.
Root rot is common on diseased tap, lateral and feeder roots, as well as on the delicate nodule tissues that contain nitrogen-fixing bacteria.
Lesions
Roots of severely diseased plants can have darkened root lesions from the crown-root junction and below.
Lesions are medium to dark brown and sometimes have a thin, darker band around the perimeter. If present, this dark band is a unique characteristic of BRR that’s considered diagnostic (Figure 1).
If environmental conditions promote disease development, lesions expand asymmetrically, eventually girdling the root.
Diseased plants can survive some root tissue loss (Figure 2), but severe lesions located on the upper tap root usually result in plant death. Good root health at this location is vital to plant survival.
Tap root
The upper 4 to 6 inches of tap root store nonstructural carbohydrates and proteins. This nutrient storage site contains the energy needed to support plant regrowth until adequate leaf tissues establish. If roots are rotted at this critical location, plants will either die or lack vigor for regrowth (Figure 3).
Severity
Conversely, if BRR symptoms don’t become severe, diseased plants will likely yield less forage than their healthy counterparts, and may recover to some extent during the growing season. However, symptoms will become increasingly severe on susceptible hosts during subsequent winters, increasing the odds of winterkill.
While BRR has been known in North America for many years, until recently, the disease was labor-intensive and time-consuming to diagnose. To overcome these difficulties, Minnesota researchers developed a rapid, sensitive assay using quantitative real-time polymerase chain reaction (qRT PCR) to identify and measure the extent of fungal colonization in plant tissues and soils.
Scientists in an increasing number of U.S. states and Canadian provinces have used this assay to identify BRR-diseased plants from commercial production fields. Ongoing research in Minnesota and elsewhere aims to understand the environmental conditions that promote BRR disease development.
Phytophthora root rot (PRR)
Phytophthora root rot (PRR) is another soil-borne disease that’s common and widespread in Minnesota.
Loss caused by PRR usually corresponds to field topography. Spread by mobile zoospores, this pathogen causes disease during wet growing seasons and in low-lying areas of the field that have had waterlogged soil for an extended period of time.
Root rot lesions appear reddish brown to tan, and are soft. When advanced, the disease can girdle and rot tap roots. As the disease progresses, PRR lesions girdle and rot root tissues in an upward direction.
This pathogen doesn’t produce observable structures on diseased plant tissues and root symptoms lack uniquely diagnostic characteristics. Commercial varieties with resistance to PRR are widely grown. This isn’t the case for BRR.
Clover root curculio larvae
Clover root curculio larvae damages young alfalfa stands when populations of the insect become increasingly large.
The grub-like curculio feeds on surface tissues of roots, leaving open, furrow-shaped wounds. Later, tap root tissues appear sunken at old larval feeding sites, much like advanced BRR lesions.
Damaged roots often have a darker brown color near feeding sites. This coloring may be the result of secondary fungal and bacterial colonizers.
Winter injury
Winter injury occurs when alfalfa varieties are challenged by severe or extended winter weather conditions beyond their winterhardiness threshold levels. Poor stand management and growing non-adapted varieties are also known to contribute to this issue.
Tap roots of winter-killed plants appear soft and pliable, with slight discoloration near the plant’s crown. Roots are much lighter than either BRR or PRR diseases. If examined soon after thawing, the root won’t be rotted.
Understanding BRR
Areas of North America
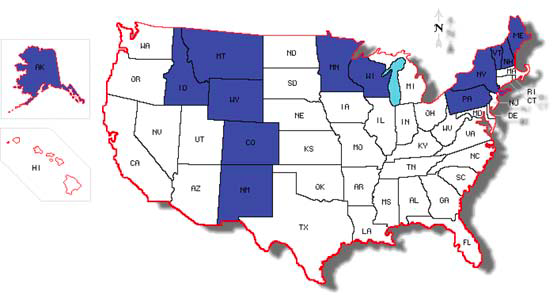
Once thought of as an insignificant alfalfa disease south of the U.S.-Canada border, recent data on BRR refute earlier assumptions.
Brown root rot has been reported in Canada from five westerly provinces, the Northwest Territories, Nova Scotia and the Yukon, as well as in the United States from Alaska and several of the 48 contiguous states.
Wyoming researchers identified it in the contiguous United States in 1996. It was detected at about the same time in Montana and later in Idaho.
In 2003, diseased plants were identified from commercial production fields in Minnesota and Wisconsin. Shortly thereafter, Minnesota researchers developed a lab assay to detect and quantify the pathogen from plant and soil samples.
BRR has now been reported from northeast locations such as New York, Maine, Pennsylvania, Vermont, New Hampshire and Ontario, Canada, as well as high-altitude locations in Colorado and New Mexico (Figure 4). This disease likely occurs in the Dakotas as well.
Areas in Minnesota and Wisconsin
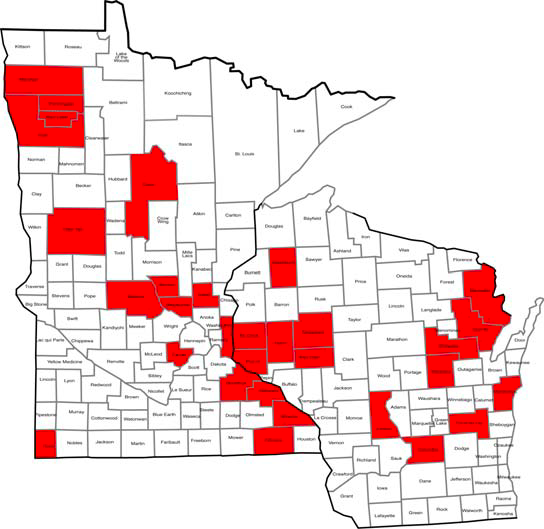
A survey of more than 100 alfalfa production fields in 38 Minnesota and 19 Wisconsin counties gathered information about areas with increased prevalence of P. sclerotioides.
About one-third of the surveyed counties had at least one field in which plants tested positive for the fungus. A higher number of fields in the following three areas were positive for the pathogen:
-
The Red River Valley in northwestern Minnesota.
-
The St. Croix River Valley in Minnesota and Wisconsin.
-
West of Green Bay in northeastern Wisconsin (Figure 5).
The pathogen is relatively widespread across the two states.
Fungal structures
Adept at surviving in northern climates, P. sclerotioides produce two fungal structures to ensure proliferation in different environments.
Shorter-term survival structures, called pycnidia, are slightly embedded in, or superficially attached to diseased plant tissues.
As small as a grain of salt, they appear gray to brownish in color and are roughly flask-shaped. One to several appendages (beaks) extend from the body of the pycnidium from which spores (conidia) are released (Figure 6).
Occasionally, pycnidia are observed on low-growing, aboveground plant tissues. It’s not clear how these fungal structures contribute to the spread of the disease.
Longer-term survival structures, called pycnosclerotia, are conidia-free and densely constructed. This structure is produced when the environment isn’t favorable for disease development. Soil-borne pycnosclerotia germinate when a host root is nearby and environmental conditions promote disease development.
Fungal growth
The fungal pathogen (P. sclerotioides) requires cool soil temperatures to cause disease.
It’s relatively dormant during the summer when soil temperatures are warm and other soilborne pathogens are most active. However, when soil temperatures cool in the autumn, P. sclerotioides become increasingly active.
Mycelial growth and pycnidia production are optimally supported at soil temperatures around 50 degrees Fahrenheit. Fungal growth can occur at lesser or greater efficiencies, in soil temperatures ranging from 32 to 81 degrees. Cooler soil temperatures – between 35 to 40 degrees – promote host infection.
It’s described as a snow mold because its growth is most rapid when soils are covered by snow.
Brown root rot occurs on a number of crops, and the fungus has been found associated with additional winter cereals, perennial grasses and biennial hollyhock. An excellent saprophyte, P. sclerotioides colonize residues from crops it attacks as well as those it doesn’t.
While a number of weedy plant species have been implicated as hosts of P. sclerotioides, tests haven’t been conducted to determine which weeds host the fungus.
Crops BRR occurs in
The fungus that causes BRR survives in diseased host tissues and by colonizing debris from crops that aren’t live hosts (saprophytic colonization).
Pathogenic and saprophytic
- Alfalfa
- Alsike clover
- Birdsfoot trefoil
- Cicer milkvetch
- Common clover
- Red clover
- Sainfoin
- Sweet clover
- Winter wheat
Saprophytic
- Barley
- Corn
- Canola
- Hairy vetch
- Oat
- Soybean
- Spring wheat
- Winter rye
Managing the disease
There are limited disease management strategies for soil-borne pathogens. Overall, strategies should focus on maintaining good plant health, particularly as the growing season draws to a close.
Like other disease issues, an integrated management approach is the best strategy to minimize the effects of BRR in your stand.
Minimizing late-season energy expenditures makes it more likely that plants will have sufficient energy to survive the winter. You can do this by avoiding excessive harvesting and allowing sufficient foliar re-growth to resupply storage organs after the final harvest and before a killing freeze.
Maintaining adequate soil nutrients in plant-available form is essential for a healthy and vigorous stand.
When an alfalfa stand’s production declines below what’s economically sustainable, rotate the field out of susceptible leguminous and grass forage species for up to three years. Avoid corn (grain as well as silage production) or soybean rotations, as Minnesota research has shown that P. sclerotioides efficiently colonize debris from both crops, surviving the winters at elevated inoculum levels.
Rotating with spring-sown small grain crops such as oat, barley and wheat reduces inoculum levels, as do fallow fields.
Peace alfalfa variety
Peace alfalfa was bred from persistent plants surviving in a BRR-infested field near Fort Vermilion in Alberta, Canada, where the disease is well-known. The variety is BRR-resistant, survives harsh, extended winters and is extremely fall dormant.
Fall dormancy
Its fall dormancy class (FDC) rating is a 1 on a scale of 1 (most dormant) to 11 (least dormant). Its performance potential in the north central United States hasn’t been tested, but it’s expected to be less favorable compared to locally adapted varieties.
The FDC system is an estimate of the amount of energy varieties partition toward the end of the growing season. The smaller the class (1 to 4), the more plant energy is placed into nonstructural nutrient reserve organs before winter.
This isn’t only beneficial for surviving extended winter periods in northerly regions, it’s also advantageous if roots become compromised by disease. Unfortunately, a consequence of growing increasingly fall-dormant varieties is that less plant energy will be partitioned to support forage production.
However, if persistence is an important production goal, less yield potential may be acceptable.
Complete listing: Alfalfa varieties and their characteristics
Chemical control
Fungicide application isn’t considered to be an effective tool for managing BRR because active ingredient activity is needed in the root tissues of mature plants. Seed treatments or foliar applications of fungicides won’t benefit diseased roots.
The pathogen causing BRR of alfalfa is widespread across Minnesota, but disease severity is closely linked with fall to spring weather conditions and stand management.
In a worst case scenario, the disease can kill plants from fall through spring. In less severe cases, diseased plants may survive and even somewhat recover depending on their susceptibility level and overall health. Managing crop stressors will help extend the productive life of your diseased alfalfa stands.
The use of trade, firm or corporation names is for your information and convenience. Its use doesn’t constitute an official endorsement or approval by the U.S. Department of Agriculture or the Agricultural Research Service.
Davidson, J.G.N. (1990). Brown root rot. In D.L. Stuteville & D.C. Erwin (Eds.), Compendium of Alfalfa Diseases (2nd ed.), St. Paul, MN: vAPS Press.
Gray, F.A., Hollingsworth, C.R., Koch, D.W., Groose, R.W., Gray, A.J., Woods, D.M., Reedy Jons, C.J., Heald, T.E., & Gill, J.R. (2007). Brown root rot of alfalfa. University of Wyoming Plant Sciences Timely Information Series Bulletin.
Hollingsworth, C.R. (2002). Assessing heritability of brown root rot (Phoma sclerotioides) resistance and forage yield in nine alfalfa (Medicago sativa ssp. sativa) populations (doctoral dissertation). University of Wyoming, Laramie.
Hollingsworth, C.R., Gray, F.A., Groose, R.W., Koch, E.W., & Heald, T.E. (2003). Distribution and incidence of brown root rot of alfalfa in Wyoming. Canadian Journal of Plant Pathology, 25, 215-217.
Larsen, J.E., Hollingsworth, C.R., Flor, J., Dornbusch, M.R., Simpson, N.L., & Samac, D.A. (2007). Distribution of Phoma sclerotioides on alfalfa and winter wheat crops in the North Central U.S. Plant Disease, 91, 551-558.
Rehm, G., Schmitt, M., & Munter, R. (2000). Fertilizing alfalfa in Minnesota. UMN Ext. bulletin FO-03814.
Samac, D.A., Miyamoto, C., Larsen, J.E., Atkinson, L., Hollingsworth, C.R., & Motteberg, C.D. (2008). Affect of crop residue on colonization and survival of Phoma sclerotioides, the causal agent of brown root rot of alfalfa. Phytopathology, 98, S139.
Sanford, G.B. (1933). A root rot of sweet clover and related crops caused by Plenodomus meliloti Dearness and Sanford. Canadian Journal of Research, 8, 337-348.
Wunsch, M.J., Dillon, M.A., Torres, R., Schwartz, H.F., & Bergstrom, G.C. (2008). First report of brown root rot of alfalfa caused by Phoma sclerotioides in Colorado and New Mexico. Plant Disease, 92, 653.
Wunsch, M.J., Kersbergen, R., Tenuta, A.U., Hall, M.H., & Bergstrom G.C. (2009). First report of brown root rot of alfalfa caused by Phoma sclerotioides in Maine, Ontario, and Pennsylvania. Plant Disease, 93, 317.
Reviewed in 2021